Sakata Group
Structural biology of protein quality control
Proteins play fundamental roles to operate cellular functions. The ubiquitin proteasome system (UPS) is essential to maintain protein homeostasis and conducts most regulated protein degradation in eukaryotes, and its malfunction leads to numerous human diseases such as cancer and neurodegenerative diseases (Hershko and Ciechanover, 1998). UPS substrates are typically first modified with chains of the small protein ubiquitin, which targets the substrate to the 26S proteasome for degradation. The 26S proteasome is a 2.5 MDa multisubunit ATP-dependent peptidase complex that consists of a barrel-shaped proteolytic 20S core particle (CP) and one or two 19S regulatory particles (RPs) that cap the CP ends (Wehmer and Sakata, 2016).
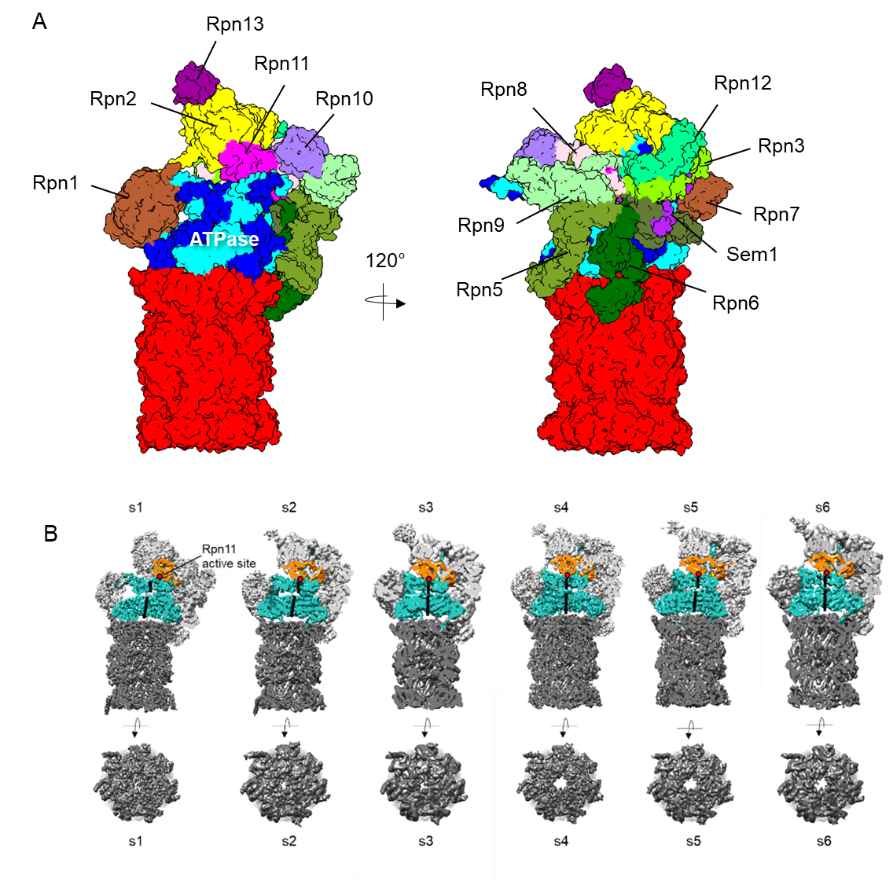
Figure 1 (A) Cryo-EM structure of the 26S proteasome (B) Conformational dynamics of the proteasome. Top views of the 20S CP are shown below. (Eisele et al., 2018; Wehmer et al., 2017)
Our research addresses the fundamental mechanisms governing protein fate by the protein quality control system, particularly protein degradation by ubiquitin-proteasome system (UPS). We focus on unraveling how the molecular machineries for protein degradation execute their function and how the AAA+ ATPases, which are the main force generator of substrate unfolding and translocation, convert chemical energy to mechanical force. We answer these questions using cryo-electron microscope (cryo-EM) single particle analysis (SPA) and other biochemical and biophysical methods, providing the structural basis for conformational dynamics and regulatory mechanisms of protein assemblies.
Our cryo-EM studies have revealed that the conformational dynamics of the 26S proteasome are tightly related to executing its substrate processing functions. Nucleotide-binding pockets of the ATPase subunits were arranged in a coordinated manner, showing that ATP hydrolysis proceeds sequentially (Eisele et al., 2018; Wehmer et al., 2017). We are seeking a deeper-understanding of the conformational dynamics of the proteasome and its regulatory mechanisms. Besides the proteasome, another AAA+ ATPase known as p97/Cdc48 plays key roles in substrate processing in the UPS. We aim to address the structural basis of the sequential substrate processing by p97 and the 26S proteasome. Our research will lead us to understand the mechanisms governing protein fate by these ATPases at the molecular and atomic levels.
My research group is part of the University Medical Center Göttingen, Institute for
Auditory Neuroscience and the Multiscale Bioimaging Excellence Cluster (MBExC),
providing us many exciting new directions and collaboration opportunities. We are going
to investigate protein homeostasis in inner ear cells where the UPS plays an important
role. In collaboration with the groups of Dr. Julia Preobraschenski and Prof. Tobias
Moser, we also aim to understand the structural basis for the function of otoferlin,
which is responsible for neurotransmitter release in inner hair cells.
References
Shein M, Hitzenberger M, Cheng TC, Rout SR, Leitl KD, Sato Y, Zacharias M, Sakata E, Schütz AK (2024) Characterizing ATP processing by the AAA+ protein p97 at the atomic level. Nat Chem 16 (3):363-372. doi: 10.1038/s41557-024-01440-0.
Hung KYS, Klumpe S, Eisele MR, Elsasser S, Tian G, Sun S, Moroco JA, Cheng TC, Joshi T, Seibel T, Van Dalen D, Feng XH, Lu Y, Ovaa H, Engen JR, Lee BH, Rudack T, Sakata E, Finley D (2022) Allosteric control of Ubp6 and the proteasome via a bidirectional switch. Nat Commun 13(1):838. doi: 10.1038/s41467-022-28186-y.
Sakata E, Eisele MR, Baumeister W (2021) Molecular and cellular dynamics of the 26S proteasome. Biochim Biophys Acta Proteins Proteom 1869(3):140583. doi: 10.1016/j.bbapap.2020.140583.
Eisele MR, Reed RG, Rudack T, Schweitzer A, Beck F, Nagy I, Pfeifer G, Plitzko JM, Baumeister W, Tomko RJ, et al. (2018) Expanded Coverage of the 26S Proteasome Conformational Landscape Reveals Mechanisms of Peptidase Gating. Cell Rep 24, 1301-1315.e1305.
Wehmer M, Rudack T, Beck F, Aufderheide A, Pfeifer G, Plitzko JM, Forster F, Schulten K, Baumeister W, Sakata E (2017) Structural insights into the functional cycle of the ATPase module of the 26S proteasome. Proc Natl Acad Sci U S A 114, 1305-1310.

