Wichmann Group
Molecular architecture of synapses
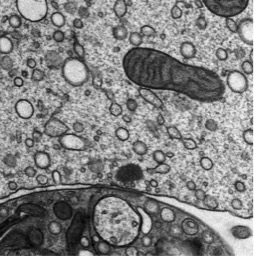
We combine immunohistochemistry, high resolution light microscopy and electron microscopy to study the molecular architecture of synapses and how structure relates to function. In general, synapses comprise several morphologically and functionally distinguishable compartments as the presynaptic cytomatrix at the active zone (CAZ) - a meshwork of proteins, essential for synaptic vesicle docking and release - and the postsynaptic density (PSD) which contains a matrix of cell-adhesion molecules, receptors and cytoskeletal elements.
The sensory inner hair cell (IHC) ribbon synapses are of particular interest for us. These synapses are characterized by an ellipsoid synaptic ribbon, which is surrounded by a halo of synaptic vesicles and has been proposed to support sustained exocytosis over long time periods. Using electron microscopic techniques as electron tomography and high-pressure freezing/freeze-substitution (HPF/FS) we study morphological aspects of wild-type and mutant synapses qualitatively and quantitatively. Electron tomography enables the visualization of fine molecular structures due to its high z-resolution. HPF/FS is a special preparation method avoiding chemical fixation of the tissue. Under high pressure and low temperature samples become immobilized within milliseconds and preserved in a near native state. The combination of electron tomography and HPF/FS allows us to determine synaptic vesicle pools, docking or tethering of synaptic vesicles under different conditions (resting, inhibited or stimulated). The activation of IHCs is achieved by using electrolyte-solutions or, taking advantage of optogenetics, by using light. With a specific high-pressure freezer equipped with an internal light source we can apply precise stimulation protocols and subsequently freeze the sample within milliseconds in order to capture ultrastructural changes with high spatio-temporal precision of different release phases at IHC ribbon synapses.
Further, we want to construct a coherent picture of the localization of synaptic proteins at IHC ribbon synapses and also the innervation pattern of afferent and efferent fibers using immunofluorescence and immunogold labeling methods in combination with high resolution light microscopy and electron tomography or serial-block-face scanning-electron-microscopy.